Lesson 8. Hepatitis B Reactivation
Associate Editor
Assistant Professor
Division of Allergy & Infectious Diseases
University of Washington
Associate Editor
Professor of Medicine
Division of Allergy & Infectious Diseases
University of Washington
Learning Objective Performance Indicators
- Recognize hepatitis B virus (HBV) reactivation, HBV flare, and HBV-associated liver failure
- Identify risk factors for HBV reactivation
- Implement HBV screening recommendations for patients at risk for HBV reactivation
- Provide HBV antiviral prophylaxis in selected individuals at risk for HBV reactivation
- Stratify risk of HBV reactivation based on hepatitis B serologic status and type of immunosuppressive therapy
Background
Hepatitis B virus (HBV) reactivation is a well-described clinical entity characterized by a rise in HBV DNA and typically followed by a rise in aminotransferase levels.[1] Although HBV reactivation can occur spontaneously or in the setting of hepatitis C virus (HCV) treatment, it is most commonly triggered by immunosuppression.[2] In addition, in persons receiving treatment for HBV, reactivation almost always occurs following withdrawal of HBV antiviral therapy. Reactivation of HBV can lead to acute hepatitis and even liver failure; these complications, however, can be prevented with the use of antiviral therapy, when indicated.
Definitions
The following summarizes the American Association for the Study of Liver Diseases (AASLD) definitions related to HBV reactivation.[3]
- Hepatitis B Reactivation: Hepatitis B virus reactivation is the loss of HBV immune control in persons who are receiving immunosuppressive therapy for a concomitant medical condition and at baseline are either (1) hepatitis B surface antigen (HBsAg) positive and anti-HBc positive or (2) HBsAg negative and anti-HBc positive.
- Among persons who are HBsAg positive and anti-HBc positive, the AASLD defines loss of immune control as constituting reactivation when:
- HBV DNA level that increases 100-fold (2-log) or greater compared to the baseline level;
- HBV DNA level is 1,000 IU/mL or greater in a person with a previously undetectable level (given that HBV-DNA levels fluctuate); or
- HBV DNA level is 10,000 IU/mL or greater if the baseline level is not available.
- Among persons who are HBsAg negative and anti-HBc positive, the AASLD defines loss of immune control as constituting reactivation when:
- There is detectable HBV DNA, or
- Reversal of HBsAg seroconversion occurs (reappearance of HBsAg).
- Among persons who are HBsAg positive and anti-HBc positive, the AASLD defines loss of immune control as constituting reactivation when:
- Hepatitis B Flare: Hepatitis B virus flares may accompany HBV reactivation and are defined as an alanine aminotransferase level (ALT) increase of at least 3 times baseline and reaching an absolute value greater than 100 U/L.
- HBV-Associated Liver Failure: HBV reactivation can result in liver failure, which the AASLD defines as at least one of the following:
- Impaired synthetic function (total bilirubin greater than 3 mg /dL or an international normalized ratio [INR] greater than 1.5);
- Ascites;
- Encephalopathy;
- Death following HBV-associated liver failure is attributed to HBV reactivation.
Pathophysiology of HBV Reactivation
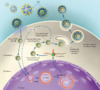
Hepatitis B virus is a partially double-stranded DNA virus.[4] During an initial HBV infection, HBV viral particles enter hepatocytes, and genomic material is brought into the nucleus, where viral genomes are converted into covalently closed circular DNA (cccDNA) molecules (Figure 1).[4,5] These cccDNA molecules serve as the template for viral mRNA and, ultimately, viral replication.[2,5] Even in individuals who have resolution of their HBV infection, cccDNA persists within hepatocytes, serving as a molecular reservoir for potential future viral reactivation and replication. Similarly, current antiviral therapies that shut down HBV replication do not eliminate HBV cccDNA. Thus, even in persons with resolved or treated HBV, the cccDNA poses a permanent risk for reactivation, particularly in clinical settings associated with immunosuppression.[6]
Clinical Features of HBV Reactivation
The reactivation of HBV can result in manifestations that range in severity from silent reactivation, which is characterized by elevated HBV DNA but no overt signs of hepatitis, to HBV-associated hepatitis (with clinical, laboratory and histological evidence of hepatitis), and to fulminant liver failure, marked by synthetic dysfunction in conjunction with clinical symptoms, including ascites and encephalopathy.[2] These clinical and laboratory manifestations are not mutually exclusive but rather exist on a continuum, with the natural history of HBV reactivation typically progressing from increased viral replication, to clinically apparent disease activity, and finally to either recovery or progression to fulminant liver failure (Figure 2).[1,7]
Risk Factors for HBV Reactivation
Persons with chronic HBV infection (HBsAg positive), as well as those with resolved HBV infection (HBsAg negative and anti-HBc positive), are at risk for reactivation of HBV. The following outlines persons with resolved, treated, or active HBV who are at risk for HBV reactivation due to immunosuppressive therapy.
- Persons Receiving Chemotherapy and Immunomodulatory Therapies: Receipt of chemotherapy, or other anticancer immunomodulatory therapy, including immune checkpoint inhibitors and tyrosine kinase inhibitors, for solid and/or liquid tumors is a well-described risk factor for HBV reactivation.[8,9,10,11] Although the risk of reactivation depends on the individual’s HBsAg status and the specific chemotherapy used, the risk of reactivation may be as high as 50 to 60% among HBsAg-positive persons who receive CD20-depleting agents without antiviral prophylaxis.[8,12]
- Individuals Receiving Treatment for Autoimmune Disease or Other Conditions Requiring Immunomodulatory Therapy: Individuals receiving immunomodulatory medications, regardless of the underlying indication, for the treatment of autoimmune disease are also at risk for HBV reactivation. Hepatitis B reactivation and flares have been described following treatment with tumor necrosis factor (TNF)-alpha inhibitors, anti-CD20 agents, corticosteroids, anti-T cell therapy, Janus kinase (JAK) inhibitors, anti-IL6 therapy, and other cytokine and integrin inhibitors.[13,14,15,16,17,18,19,20,21,22,23...] The reactivation risk is highly dependent on the baseline HBsAg status and the specific immunomodulatory medication used.[8]
- Persons Undergoing Solid Organ or Bone Marrow Transplantation: Hepatitis B reactivation has been well described among individuals who undergo solid organ or bone marrow transplantation, including among HBsAg-negative and anti-HBc-positive patients.[24,25] Hepatitis B reactivation has also been described among individuals who were previously negative for HBV but received a liver transplant from an anti-HBc positive donor.[26,27]
- Persons Receiving Treatment for HCV: Reactivation of HBV has been increasingly recognized as a potential adverse event associated with HCV direct-acting antiviral (DAA) therapy.[28,29,30,31] In this setting, HBV reactivation has been observed with multiple different DAA regimens.[28,30] Although the risk is highest for HBsAg-positive individuals receiving DAAs, rare cases of reactivation have occurred in persons with isolated anti-HBc.[28,30] The mechanism for HBV reactivation during treatment with HCV DAA treatment remains unknown.
- Chimeric Antigen Receptor (CAR) T-Cell Therapy: Chimeric antigen receptor (CAR) T cell therapy has been shown to increase the risk for HBV reactivation in patients who are HBsAg-negative and anti-HBc-positive. Based on biologic plausibility, CAR-T cell therapy is also thought to be a risk factor for HBV reactivation in HBsAg-positive persons, but all studies with this population included persons on antiviral prophylaxis to prevent HBV reactivation.[32,33]
- Transarterial Chemoembolization (TACE): TACE is a minimally invasive procedure that delivers chemotherapy agents and embolic agents directly to liver tumors and their blood supply. Although this procedure does not involve systemic chemotherapy, it can increase the risk for HBV reactivation, particularly for HBsAg-positive individuals.[34]
Assessing Risk for HBV Reactivation
When assessing the risk of HBV reactivation, it is important to consider both virologic factors and immunosuppressive factors, specifically hepatitis B serologic status and type of immunosuppression.
- HBV Serologic Status: There are two main serologic groups that indicate individuals at risk for HBV reactivation in the setting of immunosuppression: (1) HBsAg-positive and anti-HBc-positive or (2) HBsAg-negative and anti-HBc-positive.
- HBsAg-positive and anti-HBc-positive: Studies indicate that anti-HBc-positive persons are up to 7 to 8 times more likely to experience HBV reactivation if they are HBsAg-positive than if they are HBsAg-negative.[8,16,35] As an example, prior studies with anti-HBc-positive participants showed that HBV reactivation following chemotherapy occurred in 41 to 53% of HBsAg-positive persons versus 8 to 18% of those who were HBsAg-negative, and following receipt of anti-rheumatic therapies, HBV reaction occurred in 12.3% of HBsAg-positive persons versus 1.7% of those who were HBsAg-negative.[3,36,37] Persons who are HBsAg-positive and have a detectable baseline HBV DNA are at the highest risk for HBV reactivation and associated complications, based on studies performed in patients receiving cytotoxic chemotherapy and in the stem cell transplant population.[38,39,40]
- HBsAg-negative and anti-HBc-positive: The risk of reactivation is lower in HBsAg-negative and anti-HBc-positive persons when compared to HBsAg-positive and anti-HBc-positive individuals, Nonetheless, these patients remain at significant risk for HBV reactivation, particularly with the use of high-risk immunosuppressive drugs, such as B-cell depleting agents. In these settings, rates of reactivation up to 40% have been described among persons who are HBsAg negative and anti-HBc positive.[41,42,43] The presence of anti-HBs among persons who are HBsAg negative and anti-HBc positive has been shown to lower the risk of reactivation in some studies; however, data on anti-HBs titers (e.g., the anti-HBs titers needed to confer additional protection) are largely missing from the literature.[39,41,44] As such, professional society guidelines do not recommend basing antiviral prophylaxis decisions on the presence or absence of anti-HBs in persons who are HBsAg-negative and anti-HBc-positive.[3,8]
- Immunosuppressive Factors: In addition to HBsAg status, the risk of HBV reactivation depends on the immunosuppressive medication(s) received. Many different immunosuppressive medications have been linked to HBV reactivation, with B-cell depletion agents posing some of the highest risk for reactivation.[8] When multiple drugs are given concurrently or in quick succession, it is challenging to discern the precise risk posed by specific drugs due to a lack of systematically collected data.[3]
Risk Categories for HBV Reactivation
Based on studies and reports in the literature, the American Association for the Study of Liver Diseases (AASLD) and the American Gastroenterological Association (AGA) have categorized HBV serologic status and immunosuppressive medications by their level of risk for HBV reactivation. Risk categorizations differ slightly between AASLD and AGA guidelines, and both are outlined below.
The AGA Risk Categorization for HBV Reactivation
The 2025 American Gastroenterology Association (AGA) Clinical Practice Guidelines on the Prevention and Treatment of Hepatitis B Virus Reactivation in At-Risk Individuals generated the following specific risk categories for HBV reactivation.[8]
High Risk
Persons with a greater than 10% risk of HBV reactivation are classified as high risk. These individuals include select patients who are (1) HBsAg-positive and (2) HBsAg-negative / anti-HBc-positive, as outlined below.
- HBsAg-positive patients at high risk for reactivation include those receiving:
- Anthracycline derivatives (e.g., doxorubicin, epirubicin)
- Anti-TNF therapy (e.g., infliximab, adalimumab)
- Anti-IL6 therapy (e.g., tocilizumab)
- B-cell-depleting agents (e.g., rituximab, ofatumumab)
- CAR-T cell therapy
- Cytokine/integrin inhibitors (e.g., ustekinumab, secukinumab)
- Direct-acting antivirals for hepatitis C treatment (e.g., glecaprevir-pibrentasvir, sofosbuvir-velpatasvir)
- JAK inhibitors (e.g., tofacitinib, baricitinib)
- Transcatheter arterial chemoembolization (TACE) procedures
- Tyrosine kinase inhibitors (e.g., imatinib, sunitinib)
- Moderate- to high-dose corticosteroid therapy for ≥4 weeks (moderate: 10-20mg daily of prednisone or equivalent; high: >20mg daily of prednisone or equivalent)
- HBsAg-negative and anti-HBc-positive patients at high risk for reactivation include those receiving:
- B-cell-depleting agents (e.g., rituximab; ofatumumab)
Moderate Risk
Patients with a 1 to 10% risk of HBV reactivation are classified as moderate risk. These individuals include select patients who are either (1) HBsAg-positive or (2) HBsAg-negative / anti-HBc-positive, as outlined below.
- HBsAg-positive patients at moderate risk for reactivation include those receiving:
- Anti-T cell therapy (e.g., abatacept)
- Immune checkpoint inhibitors (e.g., nivolumab, pembrolizumab, ipilimumab)
- Low-dose corticosteroids therapy for ≥4 weeks (<10mg of prednisone daily or equivalent)
- HBsAg-negative / anti-HBc-positive patients at moderate risk for reactivation include those receiving:
- Anthracycline derivatives (e.g., doxorubicin, epirubicin)
- Anti-IL6 therapy (e.g., tocilizumab)
- Anti-T cell therapy (e.g., abatacept)
- CAR-T cell therapy
- Cytokine/integrin inhibitors (e.g., ustekinumab, secukinumab)
- JAK inhibitors (e.g., tofacitinib, baricitinib)
- Transcatheter arterial chemoembolization (TACE) procedures
- Tyrosine kinase inhibitors (e.g., imatinib, sunitinib)
- Moderate- to high-dose corticosteroid therapy for ≥4 weeks (moderate: 10-20 mg daily of prednisone or equivalent; high: >20mg daily of prednisone or equivalent)
Low Risk:
Persons with a less than 1% risk of HBV reactivation are classified as low risk. These individuals include select patients who are HBsAg-positive and HBsAg-negative / anti-HBc-positive, as outlined below.
- HBsAg-positive patients at low risk for reactivation include those receiving:
- Methotrexate, 6-mercaptopurine, and azathioprine
- Corticosteroid therapy for ≤1 week
- Intra-articular corticosteroid therapy
- HBsAg-negative / anti-HBc-positive patients at low risk for reactivation include those receiving:
- Anti-TNF therapy (e.g., infliximab, adalimumab)
- Direct-acting antivirals for hepatitis C treatment (e.g., glecaprevir-pibrentasvir, sofosbuvir-velpatasvir)
- Immune checkpoint inhibitors (e.g., nivolumab, pembrolizumab, ipilimumab)
- Methotrexate, 6-mercaptopurine, and azathioprine
- Low-dose corticosteroid therapy for ≥4 weeks (<10 mg of prednisone daily or equivalent)
- Corticosteroid therapy for ≤1 week
- Intra-articular corticosteroid therapy
AASLD Risk Categorization for HBV Reactivation
The AASLD has generated the following specific risk categories for HBV reactivation based on the HBV serologic status and the type of immunosuppression the individual is receiving.[45]
- Very High Risk: Persons with an estimated risk of HBV reactivation greater than 20% are deemed to be at very high risk. These persons include:
- Persons who are HBsAg positive and receiving anti-CD20 therapy (e.g. rituximab, ofatumumab)
- Persons who are HBsAg positive and undergoing hematopoietic stem cell transplantation.
- High Risk: Persons with an estimated 11 to 20% risk of HBV reactivation are deemed to be at high risk. These individuals include:
- HBsAg-positive and receiving high-dose corticosteroids (doses equivalent to 20 mg or more of prednisone daily for at least 4 weeks)
- HBsAg-positive and receiving other cytokine inhibitors other than anti-CD20 inhibitors (e.g., anti-CD52, ustekinumab).
- Moderate Risk: Persons with a 1 to 10% risk of HBV reactivation are considered to be at moderate risk, including:
- HBsAg-positive and receiving combination cytotoxic chemotherapy without corticosteroids (e.g., cisplatin-based therapy for squamous cell carcinoma and CHOP for lymphoma)
- HBsAg positive and receiving anti-tumor necrosis factor agents
- HBsAg positive and receiving anti-rejection therapy for solid organ transplant recipients
- HBsAg negative and anti-HBc positive and receiving anti-CD20 agents
- HBsAg negative and anti-HBc positive and undergoing hematopoietic stem cell transplantation
- Low Risk: Persons with a less than 1% risk of HBV reactivation are considered low risk. This includes individuals who are:
- HBsAg positive and receiving methotrexate or azathioprine
- HBsAg negative and anti-HBc positive and receiving high-dose corticosteroids (doses equivalent to 20 mg or greater of prednisone daily for at least 4 weeks)
- HBsAg negative and anti-HBc positive and receiving other cytokine inhibitors (e.g., anti-CD52, ustekinumab).
- Rare / Very Low Risk: HBV reactivation is felt to be rare in the following groups:
- Persons who are HBsAg negative and anti-HBc positive and are receiving combination cytotoxic chemotherapy without corticosteroids
- Persons who are HBsAg negative and anti-HBc positive and are receiving anti-tumor necrosis factor agents
- Persons who are HBsAg negative and anti-HBc positive and are receiving anti-rejection therapy for solid organ transplant recipients
- Persons who are HBsAg negative and anti-HBc positive and are receiving methotrexate or azathioprine
- No Known Effect: There is no known risk of HBV reactivation associated with androgen deprivation therapy or estrogen and progesterone blockers for persons who are (1) HBsAg positive and anti-HBc positive or (2) HBsAg negative and anti-HBc positive.
HBV Screening Prior to Initiation of Immunosuppression
Given the risk for HBV reactivation among persons receiving immunosuppressive medications, several professional guidelines recommend routine HBV screening prior to the initiation of immunosuppressive therapy.[45,46] The 2025 AGA Clinical Practice Guidelines on the Prevention and Treatment of Hepatitis B Virus Reactivation in At-Risk Individuals specifically recommends screening with HBsAg, anti-HBs, and total anti-HBc, in concordance with universal Centers for Disease Control and Prevention (CDC) HBV screening recommendations, prior to initiation of immunosuppressive therapy.[8]
Data on Antiviral Therapy to Prevent HBV Reactivation
Several studies have shown that antiviral therapy can decrease the risk of HBV reactivation when initiated prior to or concurrently with immunosuppressive therapy.[34,36,47,48,49] Data from randomized clinical trials of HBsAg-positive patients undergoing chemotherapy indicate the risk of reactivation can be significantly decreased with the use of preemptive antiviral therapy.[34,36,37,49,50] In a meta-analysis of 12 randomized clinical trials comparing antiviral prophylaxis versus no prophylaxis in patients who were either HBsAg positive or anti-HBc positive / HBsAg negative, antiviral prophylaxis was associated with an 82% decrease in the risk for reactivation and a 72% decrease in the risk of hepatitis flare from HBV reactivation when compared to no prophylaxis.[8]
- Delayed vs. Preemptive Antiviral Therapy to Prevent Reactivation: In a small study of 30 HBsAg-positive persons undergoing treatment for lymphoma, investigators randomized adults to receive preemptive lamivudine 100 mg daily starting 1 week before chemotherapy versus no preemptive treatment.[36] Over the course of the study, 53% of the participants in the control arm versus 0% of those in the intervention arm experienced HBV reactivation.[36]
- Reduced Risk of Reactivation Among Persons Receiving Antiviral Prophylaxis in HBsAg-positive Persons: In a meta-analysis of 26 studies that included persons with chronic or resolved HBV who were receiving chemotherapy for a solid tumor, 13 studies compared reactivation risk among HBsAg-positive patients receiving antiviral prophylaxis versus no prophylaxis.[9] The pooled odds ratio for reactivation was 0.12 (95% CI: 0.06 – 0.22), suggesting a substantial reduction in HBV reactivation among those who received prophylactic antivirals.[9]
- Antiviral Therapy in HBsAg-negative / anti-HBc-Positive Persons: Clinical trial data similarly support the use of antiviral prophylaxis in patients with resolved HBV (HBsAg-negative / anti-HBc-positive) who are at high or moderate risk for HBV reactivation. In a randomized controlled trial of prophylactic entecavir versus no therapy among HBsAg-negative / anti-HBc-positive persons receiving chemotherapy for CD20+ lymphoma, the cumulative rate of HBV reactivation 18 months after chemotherapy was 25.9% in the control arm versus 4.3% in the entecavir arm.[37] Similarly, in a phase IV, randomized, prospective, open-label trial comparing tenofovir DF versus close monitoring in HBsAg-negative / anti-HBc-positive patients receiving rituximab, reactivation of HBV occurred in 0% of those on tenofovir DF versus 10.7% of those in the monitoring group.[50]
Recommendations for HBV Prophylactic Antiviral Therapy
The American Association for the Study of Liver Disease (AASLD), the American Gastroenterological Association (AGA), and the American Society of Clinical Oncology (ASCO) have all released recommendations on the use of antiviral prophylaxis to prevent HBV reactivation during immunosuppressive therapy, which are summarized below.[8,45,51,52]
AASLD Recommendations
The AASLD recommendations for antiviral prophylaxis to prevent HBV reactivation were published in 2015 and are based on the HBsAg and anti-HBc status of the individual under consideration and the type of immunosuppressive therapy.[45]
- HBsAg-Positive and anti-HBc-Positive: The AASLD recommends initiation of antiviral therapy in all HBsAg-positive patients prior to or at the initiation of chemotherapy, immunosuppressive therapy, hematopoietic stem cell transplant, or solid organ transplant.
- HBsAg-Negative and anti-HBc-Positive: The AASLD recommends routine antiviral prophylaxis for HBsAg-negative and anti-HBc-positive patients receiving anti-CD20 therapies and those undergoing hematopoietic stem cell transplantation. Monitoring of HBV DNA at 1- to 3-month intervals, with initiation of antiviral therapy if HBV DNA becomes detectable, is reasonable with HBsAg-negative and anti-HBc-positive patients with lesser degrees of immunosuppression.
AGA Recommendations
The AGA has generated risk group categories based on the risk of HBV reactivation.[8,51]
- High Risk: For individuals at high risk of reactivation (greater than 10%), as outlined above, the AGA recommends antiviral prophylaxis over monitoring (strong recommendation). Antiviral prophylaxis should be started before the introduction of immunosuppressive therapy.
- Moderate Risk: For individuals at moderate risk of reactivation (1–10%), as outlined above, the AGA recommends antiviral prophylaxis over monitoring alone (conditional, moderate certainty recommendation). With this recommendation, the guidelines similarly state that patients who place a higher priority on avoiding long-term use of antivirals, particularly those who are HBsAg-negative, may reasonably elect active monitoring if close follow-up and adherence to monitoring is feasible for them. If monitoring is selected as an alternative strategy, laboratory evaluation for HBV DNA and liver function tests should be performed at 1- to-3-month intervals.[8]
- Low Risk: The AGA recommends not using routine antiviral prophylaxis for patients at low risk (less than 1%) for HBV reactivation, as outlined above. This recommendation assumes the ability to perform continued monitoring based on adequate patient follow-up. Alternatively, for patients who strongly wish to avoid the small risk of reactivation, initiation of antiviral prophylaxis may be reasonable.[8]
The AGA recommendations for prophylaxis to prevent HBV reactivation were updated in 2025 and are based on the risk stratification groups listed above (Table 1).[8,51]
ASCO Recommendations
The ASCO recommendations for preventing HBV reactivation with antiviral therapy were updated in 2020 and include three categories based on the HBsAg and anti-HBc status and risk related to immunosuppression.[52]
- HBsAg-Positive and anti-HBc-Positive: The ASCO recommends initiation of antiviral therapy for all HBsAg-positive patients receiving any systemic anticancer therapy, except for isolated hormonal therapy (e.g., without systemic anticancer therapy).
- HBsAg-Negative and anti-HBc-Positive on High-Risk Immunosuppression: The ASCO recommends initiation of antiviral therapy for HBsAg-negative and anti-HBc-positive patients receiving high-risk immunosuppressive therapies, including anti-CD20 monoclonal antibodies or stem-cell transplantation. An alternative option in this group is close monitoring with HBsAg and HBV DNA every 3 months, in patients who are able to adhere to frequent and consistent follow-up, with immediate initiation of antiviral therapy if reactivation occurs.
- HBsAg-Negative and anti-HBc-Positive on Lower-Risk Immunosuppression: The ASCO recommends HBsAg-negative and anti-HBc-positive patients on lower risk immunosuppression (e.g. regimens that do not include anti-CD20 agents or stem cell transplantation) be monitored with HBsAg and ALT testing every 3 months, with subsequent HBV DNA testing should a hepatitis flare occur. Individuals with emergent evidence of HBV reactivation should be started on antiviral therapy at that time.
Choice of Antiviral Agents for the Prevention of HBV Reactivation
The AASLD and AGA recommend using either entecavir or tenofovir, two drugs with high potency and a high barrier to resistance, for the prevention of HBV reactivation.[3,8] Note, for the use of tenofovir, these guidelines do not specify the use of tenofovir DF versus tenofovir alafenamide. The use of lamivudine is not recommended due to the high rates of drug resistance that have been observed with lamivudine therapy for persons with chronic HBV. The recommendations for using entecavir or tenofovir to prevent HBV reactivation are supported by several meta-analyses and one randomized, open-label trial of entecavir versus lamivudine for the prevention of HBV reactivation in persons undergoing treatment for diffuse large B-cell lymphoma.[8,48,53,54,55] In the randomized, open-label trial, investigators observed a lower incidence of HBV-related hepatitis, HBV reactivation, and chemotherapy interruption in those receiving entecavir when compared to those receiving lamivudine for HBV prophylaxis.[37] Although there are no trials that directly compare lamivudine with tenofovir DF or tenofovir alafenamide for the prevention of HBV reactivation, tenofovir DF was found to be highly effective in preventing HBV reactivation among anti-persons who are anti-HBc positive and HBsAg negative and receiving rituximab.[8] Expert opinion supports the use of tenofovir DF or tenofovir alafenamide for the prevention of HBV reactivation, owing to its high potency and high barrier to resistance.[3,8]
Duration of Antiviral Prophylaxis
The duration of antiviral prophylaxis for the prevention of HBV reactivation is not well studied. Experts agree that antiviral therapy should be continued for the duration of immunosuppressive therapy; however, there is some uncertainty as to how long antiviral therapy should be continued after discontinuation of immunosuppression. In most studies, prophylactic therapy has been continued for 3 to 12 months following discontinuation of immunosuppressive therapy,[45,55,56] and reactivation has rarely been reported greater than 12 months following cessation of chemotherapy.[57,58] The AASLD and AGA recommend continuing antiviral therapy for at least 6 months after completion of most immunosuppressive therapy.[3,8,51] This recommendation is extended to 12 months for patients receiving B-cell-depleting agents.[3,8,51] Conversely, the ASCO recommends antiviral prophylaxis for at least 12 months following completion of all chemotherapy.[52]
Monitoring Low-Risk Patients Off Antiviral Therapy
Patients on immunomodulatory therapy who are at low risk for HBV reactivation and not initiated on prophylactic antivirals should be monitored routinely for HBV reactivation. In these patients, the AASLD and AGA recommend obtaining HBV DNA levels and ALT levels every 1 to 3 months.[3] The ASCO recommends monitoring HBsAg and ALT in these patients every 3 months while they are receiving chemotherapy.[52] How long to continue monitoring after the cessation of immunosuppressive therapy is not clear in this lower-risk group.
Special Populations to Consider for HBV Reactivation
Persons Receiving Treatment for Hepatitis C Virus Infection
Individuals with HBV and HCV coinfection are at elevated risk of HBV reactivation during HCV treatment with direct-acting antiviral (DAA) therapy.[3,28,30,59,60,61] In one meta-analysis, among HBsAg-positive individuals receiving DAA HCV therapy, the estimated risk of HBV reactivation was 14.1%, and the risk of clinical hepatitis was 12.2%.[62] In this same meta-analysis, the estimated incidence of clinical hepatitis due to HBV reactivation was 12.2% among persons receiving DAA therapy who were HBsAg-positive.[62] During HCV treatment, the risk of reactivation is highest among HBsAg-positive individuals, but rare cases have been described in HBsAg-negative and anti-HBc-positive patients.[60,63] Concurrent treatment with HBV antivirals can prevent reactivation, and in a randomized controlled trial of noncirrhotic patients with HCV and HBV coinfection assigned to either 12 weeks of DAA therapy alone versus 12 weeks of DAAs plus 12 weeks of entecavir versus 12 weeks of DAAs plus 24 weeks of entecavir, HBV reactivation occurred in 50% of those on DAAs alone and 0% of those who received concurrent entecavir for either 12 of 24 weeks.[31]
- AGA Guidance: In their updated guidance, the AGA recommends concurrent initiation of HBV prophylaxis in persons who are HBsAg positive and will be initiating DAA therapy for the treatment of HCV.[8] For persons who are anti-HBc positive and HBsAg negative, the AGA recommends monitoring over antiviral prophylaxis.[8]
- AASLD Guidance: The AASLD recommends initiating HBV antiviral therapy concurrently with DAA therapy only in persons with HBV-HCV coinfection who have cirrhosis and in those who otherwise meet AASLD guidelines for initiation of HBV antiviral therapy.[3] The AASLD states that HBsAg-positive patients who do not meet AASLD HBV treatment guideline criteria for the initiation of antiviral therapy may be monitored with HBV DNA levels every 4 to 8 weeks while on HCV DAA therapy and for 3 months post-HCV treatment.[3]
Although practice patterns vary, many experts choose to initiate HBV prophylaxis with entecavir, tenofovir DF, or tenofovir alafenamide concurrent with DAA therapy in all HBsAg-positive patients. Persons with chronic HCV infection who are HBsAg negative and anti-HBc positive have a very low risk of HBV reactivation during HCV treatment and can be monitored with ALT levels at baseline, at the end of HCV treatment, and at post-treatment follow-up.
Persons with HIV
Progressive immunodeficiency from poorly controlled HIV can lead to reactivation of HBV in persons with chronic HBV infection and can even lead to reverse seroconversion in HBV patients who recovered from HBV infection (e.g., HBsAg negative and anti-HBc positive) through a gradual loss of anti-HBs and re-emergence of HBsAg with ALT elevation.[1,64,65,66,67,68] In addition, many antiviral medications commonly used to treat or prevent HIV (tenofovir DF, tenofovir alafenamide, lamivudine, and emtricitabine) also have activity against HBV, and HBV flares can occur when these antiretroviral medications with HBV activity are discontinued.[1,69]
Summary Points
- Reactivation of HBV most often occurs in persons with underlying immunosuppression or in those who initiate immunosuppressive medications; however, it has also been described in individuals receiving direct-acting anti27811836viral therapy for HCV and those undergoing TACE procedures.
- HBV reactivation can result in manifestations that range from silent reactivation, to HBV-associated hepatitis, to fulminant liver failure.
- Persons with chronic HBV infection (HBsAg positive), as well as those with resolved HBV infection (HBsAg negative and anti HBc positive), are at risk for reactivation of HBV, with risk depending both on an individual's HBV serologic status, and their type of immunosuppression.
- Persons who are HBsAg positive and receiving anti-CD20 therapy (e.g., rituximab, ofatumumab), as well as persons who are HBsAg positive and undergoing hematopoietic stem cell transplantation, are at the highest risk for HBV reactivation.
- The CDC recommends screening for HBV in persons receiving or needing cytotoxic or immunosuppressive therapy, including chemotherapy for malignant diseases, immunosuppression related to organ transplantation, and immunosuppression for rheumatologic and gastrointestinal disorders.
- Antiviral prophylaxis should generally be initiated in HBsAg-positive patients prior to or at the initiation of chemotherapy, many immunosuppressive therapies, hematopoietic stem cell transplant, solid organ transplant, or DAA therapy for HCV.
- In general, HBsAg-negative and anti-HBc-positive patients receiving high-risk immunosuppression (e.g., anti-CD20 therapies, hematopoietic stem cell transplantation) should also be started on antiviral prophylaxis.
- Entecavir, tenofovir DF, or tenofovir alafenamide should be used if HBV prophylaxis is needed in the setting of immunosuppression.
- Antiviral therapy should be continued for the duration of immunosuppressive therapy, and in general, for 6 to 12 months following discontinuation of immunosuppressive therapy.
- Patients on immunomodulatory therapy who are at low risk for HBV reactivation and not initiated on prophylactic antivirals should be monitored routinely for HBV reactivation. In these patients, it is recommended to obtain HBV DNA levels and ALT levels every 1 to 3 months.
Citations
- 1.Hoofnagle JH. Reactivation of hepatitis B. Hepatology. 2009;49:S156-65.[PubMed Abstract] -
- 2.Myint A, Tong MJ, Beaven SW. Reactivation of Hepatitis B Virus: A Review of Clinical Guidelines. Clin Liver Dis (Hoboken). 2020;15:162-7.[PubMed Abstract] -
- 3.Terrault NA, Lok ASF, McMahon BJ, et al. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology. 2018;67:1560-99.[PubMed Abstract] -
- 4.Wei L, Ploss A. Hepatitis B virus cccDNA is formed through distinct repair processes of each strand. Nat Commun. 2021;12:1591.[PubMed Abstract] -
- 5.Ganem D, Prince AM. Hepatitis B virus infection--natural history and clinical consequences. N Engl J Med. 2004;350:1118-29.[PubMed Abstract] -
- 6.Martinez MG, Boyd A, Combe E, Testoni B, Zoulim F. Covalently closed circular DNA: The ultimate therapeutic target for curing HBV infections. J Hepatol. 2021;75:706-17.[PubMed Abstract] -
- 7.Loomba R, Liang TJ. Hepatitis B Reactivation Associated With Immune Suppressive and Biological Modifier Therapies: Current Concepts, Management Strategies, and Future Directions. Gastroenterology. 2017;152:1297-1309.[PubMed Abstract] -
- 8.Ali FS, Nguyen MH, Hernaez R, et al. AGA Clinical Practice Guideline on the Prevention and Treatment of Hepatitis B Virus Reactivation in At-Risk Individuals. Gastroenterology. 2025;168:267-84.[PubMed Abstract] -
- 9.Paul S, Saxena A, Terrin N, Viveiros K, Balk EM, Wong JB. Hepatitis B Virus Reactivation and Prophylaxis During Solid Tumor Chemotherapy: A Systematic Review and Meta-analysis. Ann Intern Med. 2016;164:30-40.[PubMed Abstract] -
- 10.Lee PC, Chao Y, Chen MH, et al. Risk of HBV reactivation in patients with immune checkpoint inhibitor-treated unresectable hepatocellular carcinoma. J Immunother Cancer. 2020;8(2):e001072.[PubMed Abstract] -
- 11.Zhang X, Zhou Y, Chen C, et al. Hepatitis B virus reactivation in cancer patients with positive Hepatitis B surface antigen undergoing PD-1 inhibition. J Immunother Cancer. 2019;7:322.[PubMed Abstract] -
- 12.Tsutsumi Y, Yamamoto Y, Ito S, et al. Hepatitis B virus reactivation with a rituximab-containing regimen. World J Hepatol. 2015;7:2344-51.[PubMed Abstract] -
- 13.Lee YH, Bae SC, Song GG. Hepatitis B virus (HBV) reactivation in rheumatic patients with hepatitis core antigen (HBV occult carriers) undergoing anti-tumor necrosis factor therapy. Clin Exp Rheumatol. 2013;31:118-21.[PubMed Abstract] -
- 14.Lee YH, Bae SC, Song GG. Hepatitis B virus reactivation in HBsAg-positive patients with rheumatic diseases undergoing anti-tumor necrosis factor therapy or DMARDs. Int J Rheum Dis. 2013;16:527-31.[PubMed Abstract] -
- 15.Navarro R, Vilarrasa E, Herranz P, et al. Safety and effectiveness of ustekinumab and antitumour necrosis factor therapy in patients with psoriasis and chronic viral hepatitis B or C: a retrospective, multicentre study in a clinical setting. Br J Dermatol. 2013;168:609-16.[PubMed Abstract] -
- 16.Pérez-Alvarez R, Díaz-Lagares C, García-Hernández F, et al. Hepatitis B virus (HBV) reactivation in patients receiving tumor necrosis factor (TNF)-targeted therapy: analysis of 257 cases. Medicine (Baltimore). 2011;90:359-71.[PubMed Abstract] -
- 17.Ryu HH, Lee EY, Shin K, et al. Hepatitis B virus reactivation in rheumatoid arthritis and ankylosing spondylitis patients treated with anti-TNFα agents: a retrospective analysis of 49 cases. Clin Rheumatol. 2012;31:931-6.[PubMed Abstract] -
- 18.Kuo MH, Tseng CW, Lu MC, et al. Risk of Hepatitis B Virus Reactivation in Rheumatoid Arthritis Patients Undergoing Tocilizumab-Containing Treatment. Dig Dis Sci. 2021;66:4026-34.[PubMed Abstract] -
- 19.Pauly MP, Tucker LY, Szpakowski JL, et al. Incidence of Hepatitis B Virus Reactivation and Hepatotoxicity in Patients Receiving Long-term Treatment With Tumor Necrosis Factor Antagonists. Clin Gastroenterol Hepatol. 2018;16:1964-1973.e1.[PubMed Abstract] -
- 20.Ting SW, Chen YC, Huang YH. Risk of Hepatitis B Reactivation in Patients with Psoriasis on Ustekinumab. Clin Drug Investig. 2018;38:873-80.[PubMed Abstract] -
- 21.Wang YH, Liang JD, Sheng WH, Tien FM, Chen CY, Tien HF. Hepatitis B reactivation during treatment of tyrosine kinase inhibitors-Experience in 142 adult patients with chronic myeloid leukemia. Leuk Res. 2019;81:95-7.[PubMed Abstract] -
- 22.Wong GL, Wong VW, Yuen BW, et al. Risk of hepatitis B surface antigen seroreversion after corticosteroid treatment in patients with previous hepatitis B virus exposure. J Hepatol. 2020;72:57-66.[PubMed Abstract] -
- 23.Zhong Z, Liao W, Dai L, et al. Average corticosteroid dose and risk for HBV reactivation and hepatitis flare in patients with resolved hepatitis B infection. Ann Rheum Dis. 2022:584-91.[PubMed Abstract] -
- 24.Hammond SP, Borchelt AM, Ukomadu C, Ho VT, Baden LR, Marty FM. Hepatitis B virus reactivation following allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2009;15:1049-59.[PubMed Abstract] -
- 25.Mei T, Noguchi H, Hisadome Y, et al. Hepatitis B virus reactivation in kidney transplant patients with resolved hepatitis B virus infection: Risk factors and the safety and efficacy of preemptive therapy. Transpl Infect Dis. 2020;22:e13234.[PubMed Abstract] -
- 26.Khiangte B, Kothakota SR, Sasidharan M, et al. Hepatitis B Reactivation in Liver Transplant Recipients With Hepatitis B Virus Core Antibody Positive Grafts: a Retrospective Study. J Clin Exp Hepatol. 2020;10:548-54.[PubMed Abstract] -
- 27.Wei L, Chen D, Zhang B, et al. Long-term outcome and recurrence of hepatitis B virus following liver transplantation from hepatitis B surface antigen-positive donors in a Chinese population. J Viral Hepat. 2018;25:1576-81.[PubMed Abstract] -
- 28.Wang C, Ji D, Chen J, et al. Hepatitis due to Reactivation of Hepatitis B Virus in Endemic Areas Among Patients With Hepatitis C Treated With Direct-acting Antiviral Agents. Clin Gastroenterol Hepatol. 2017;15:132-6.[PubMed Abstract] -
- 29.Ou P, Fang Z, Chen J. Hepatitis B reactivation in a chronic hepatitis C patient treated with ledipasvir and sofosbuvir: A case report. Clin Res Hepatol Gastroenterol. 2017;41:e17-e18.[PubMed Abstract] -
- 30.Bersoff-Matcha SJ, Cao K, Jason M, et al. Hepatitis B Virus Reactivation Associated With Direct-Acting Antiviral Therapy for Chronic Hepatitis C Virus: A Review of Cases Reported to the U.S. Food and Drug Administration Adverse Event Reporting System. Ann Intern Med. 2017;166:792-8.[PubMed Abstract] -
- 31.Cheng PN, Liu CJ, Chen CY, et al. Entecavir Prevents HBV Reactivation During Direct Acting Antivirals for HCV/HBV Dual Infection: A Randomized Trial. Clin Gastroenterol Hepatol. 2022;20:2800-8.[PubMed Abstract] -
- 32.Li P, Zhou L, Ye S, et al. Risk of HBV Reactivation in Patients With Resolved HBV Infection Receiving Anti-CD19 Chimeric Antigen Receptor T Cell Therapy Without Antiviral Prophylaxis. Front Immunol. 2021;12:638678.[PubMed Abstract] -
- 33.Wang Y, Liu Y, Tan X, et al. Safety and efficacy of chimeric antigen receptor (CAR)-T-cell therapy in persons with advanced B-cell cancers and hepatitis B virus-infection. Leukemia. 2020;34:2704-7.[PubMed Abstract] -
- 34.Jang JW, Choi JY, Bae SH, et al. A randomized controlled study of preemptive lamivudine in patients receiving transarterial chemo-lipiodolization. Hepatology. 2006;43:233-40.[PubMed Abstract] -
- 35.Shouval D, Shibolet O. Immunosuppression and HBV reactivation. Semin Liver Dis. 2013;33:167-77.[PubMed Abstract] -
- 36.Lau GK, Yiu HH, Fong DY, et al. Early is superior to deferred preemptive lamivudine therapy for hepatitis B patients undergoing chemotherapy. Gastroenterology. 2003;125:1742-9.[PubMed Abstract] -
- 37.Huang YH, Hsiao LT, Hong YC, et al. Randomized controlled trial of entecavir prophylaxis for rituximab-associated hepatitis B virus reactivation in patients with lymphoma and resolved hepatitis B. J Clin Oncol. 2013;31:2765-72.[PubMed Abstract] -
- 38.Lau GK, Leung YH, Fong DY, et al. High hepatitis B virus (HBV) DNA viral load as the most important risk factor for HBV reactivation in patients positive for HBV surface antigen undergoing autologous hematopoietic cell transplantation. Blood. 2002;99:2324-30.[PubMed Abstract] -
- 39.Yeo W, Zee B, Zhong S, et al. Comprehensive analysis of risk factors associating with Hepatitis B virus (HBV) reactivation in cancer patients undergoing cytotoxic chemotherapy. Br J Cancer. 2004;90:1306-11.[PubMed Abstract] -
- 40.An J, Shim JH, Kim SO, et al. Comprehensive outcomes of on- and off-antiviral prophylaxis in hepatitis B patients undergoing cancer chemotherapy: A competing risks analysis. J Med Virol. 2016;88:1576-86.[PubMed Abstract] -
- 41.Hsu C, Tsou HH, Lin SJ, et al. Chemotherapy-induced hepatitis B reactivation in lymphoma patients with resolved HBV infection: a prospective study. Hepatology. 2014;59:2092-100.[PubMed Abstract] -
- 42.Seto WK, Chan TS, Hwang YY, et al. Hepatitis B reactivation in occult viral carriers undergoing hematopoietic stem cell transplantation: A prospective study. Hepatology. 2017;65:1451-61.[PubMed Abstract] -
- 43.Seto WK, Chan TS, Hwang YY, et al. Hepatitis B reactivation in patients with previous hepatitis B virus exposure undergoing rituximab-containing chemotherapy for lymphoma: a prospective study. J Clin Oncol. 2014;32:3736-43.[PubMed Abstract] -
- 44.Pei SN, Ma MC, Wang MC, et al. Analysis of hepatitis B surface antibody titers in B cell lymphoma patients after rituximab therapy. Ann Hematol. 2012;91:1007-12.[PubMed Abstract] -
- 45.Di Bisceglie AM, Lok AS, Martin P, Terrault N, Perrillo RP, Hoofnagle JH. Recent US Food and Drug Administration warnings on hepatitis B reactivation with immune-suppressing and anticancer drugs: just the tip of the iceberg? Hepatology. 2015;61:703-11.[PubMed Abstract] -
- 46.European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatitis B virus infection. J Hepatol. 2025:S0168-8278(25)00174-6.[PubMed Abstract] -
- 47.Long M, Jia W, Li S, et al. A single-center, prospective and randomized controlled study: Can the prophylactic use of lamivudine prevent hepatitis B virus reactivation in hepatitis B s-antigen seropositive breast cancer patients during chemotherapy? Breast Cancer Res Treat. 2011;127:705-12.[PubMed Abstract] -
- 48.Huang H, Li X, Zhu J, et al. Entecavir vs lamivudine for prevention of hepatitis B virus reactivation among patients with untreated diffuse large B-cell lymphoma receiving R-CHOP chemotherapy: a randomized clinical trial. JAMA. 2014;312:2521-30.[PubMed Abstract] -
- 49.Hsu C, Hsiung CA, Su IJ, et al. A revisit of prophylactic lamivudine for chemotherapy-associated hepatitis B reactivation in non-Hodgkin's lymphoma: a randomized trial. Hepatology. 2008;47:844-53.[PubMed Abstract] -
- 50.Buti M, Manzano ML, Morillas RM, et al. Correction: Randomized prospective study evaluating tenofovir disoproxil fumarate prophylaxis against hepatitis B virus reactivation in anti-HBc-positive patients with rituximab-based regimens to treat hematologic malignancies: The Preblin study. PLoS One. 2018;13:e0199926.[PubMed Abstract] -
- 51.Reddy KR, Beavers KL, Hammond SP, Lim JK, Falck-Ytter YT. American Gastroenterological Association Institute guideline on the prevention and treatment of hepatitis B virus reactivation during immunosuppressive drug therapy. Gastroenterology. 2015;148:215-9.[PubMed Abstract] -
- 52.Hwang JP, Feld JJ, Hammond SP, et al. Hepatitis B Virus Screening and Management for Patients With Cancer Prior to Therapy: ASCO Provisional Clinical Opinion Update. J Clin Oncol. 2020;38:3698-3715.[PubMed Abstract] -
- 53.Yang C, Qin B, Yuan Z, Chen L, Zhou HY. Meta-analysis of prophylactic entecavir or lamivudine against hepatitis B virus reactivation. Ann Hepatol. 2016;15:501-11.[PubMed Abstract] -
- 54.Yu S, Luo H, Pan M, et al. Comparison of entecavir and lamivudine in preventing HBV reactivation in lymphoma patients undergoing chemotherapy: a meta-analysis. Int J Clin Pharm. 2016;38:1035-43.[PubMed Abstract] -
- 55.Zhang MY, Zhu GQ, Shi KQ, et al. Systematic review with network meta-analysis: Comparative efficacy of oral nucleos(t)ide analogues for the prevention of chemotherapy-induced hepatitis B virus reactivation. Oncotarget. 2016;7:30642-58.[PubMed Abstract] -
- 56.Liu WP, Wang XP, Zheng W, et al. Hepatitis B virus reactivation after withdrawal of prophylactic antiviral therapy in patients with diffuse large B cell lymphoma. Leuk Lymphoma. 2016;57:1355-62.[PubMed Abstract] -
- 57.Cerva C, Colagrossi L, Maffongelli G, et al. Persistent risk of HBV reactivation despite extensive lamivudine prophylaxis in haematopoietic stem cell transplant recipients who are anti-HBc-positive or HBV-negative recipients with an anti-HBc-positive donor. Clin Microbiol Infect. 2016;22:946.e1-946.e8.[PubMed Abstract] -
- 58.Nakaya A, Fujita S, Satake A, et al. Delayed HBV reactivation in rituximab-containing chemotherapy: How long should we continue anti-virus prophylaxis or monitoring HBV-DNA? Leuk Res. 2016;50:46-9.[PubMed Abstract] -
- 59.Liu CJ, Chuang WL, Sheen IS, et al. Efficacy of Ledipasvir and Sofosbuvir Treatment of HCV Infection in Patients Coinfected With HBV. Gastroenterology. 2018;154:989-97.[PubMed Abstract] -
- 60.Belperio PS, Shahoumian TA, Mole LA, Backus LI. Evaluation of hepatitis B reactivation among 62,920 veterans treated with oral hepatitis C antivirals. Hepatology. 2017;66:27-36.[PubMed Abstract] -
- 61.Park YJ, Yi KY, Woo HY, Heo J, Song GA. Risk of HBV reactivation in HBV/HCV-co-infected HCV-treated patients: A single-center study. PLoS One. 2025;20:e0324019.[PubMed Abstract] -
- 62.Chen G, Wang C, Chen J, et al. Hepatitis B reactivation in hepatitis B and C coinfected patients treated with antiviral agents: A systematic review and meta-analysis. Hepatology. 2017;66:13-26.[PubMed Abstract] -
- 63.Suda T, Shimakami T, Shirasaki T, et al. Reactivation of hepatitis B virus from an isolated anti-HBc positive patient after eradication of hepatitis C virus with direct-acting antiviral agents. J Hepatol. 2017;67:1108-11.[PubMed Abstract] -
- 64.Biggar RJ, Goedert JJ, Hoofnagle J. Accelerated loss of antibody to hepatitis B surface antigen among immunodeficient homosexual men infected with HIV. N Engl J Med. 1987;316:630-1.[PubMed Abstract] -
- 65.Lazizi Y, Grangeot-Keros L, Delfraissy JF, et al. Reappearance of hepatitis B virus in immune patients infected with the human immunodeficiency virus type 1. J Infect Dis. 1988;158:666-7.[PubMed Abstract] -
- 66.Thio CL. Hepatitis B and human immunodeficiency virus coinfection. Hepatology. 2009;49:S138-45.[PubMed Abstract] -
- 67.Vento S, Di Perri G, Garofano T, Concia E, Bassetti D. Reactivation of hepatitis B in AIDS. Lancet. 1989;2:108-9.[PubMed Abstract] -
- 68.Zhou K, Terrault N. Management of hepatitis B in special populations. Best Pract Res Clin Gastroenterol. 2017;31:311-20.[PubMed Abstract] -
- 69.Altfeld M, Rockstroh JK, Addo M, et al. Reactivation of hepatitis B in a long-term anti-HBs-positive patient with AIDS following lamivudine withdrawal. J Hepatol. 1998;29:306-9.[PubMed Abstract] -
Additional References
- American Gastroenterological Association. AGA Institute Guidelines on Hepatitis B Reactivation (HBVr): Clinical Decision Support Tool. Gastroenterology. 2015;148:220.[AGA] -
- Chen XQ, Peng JW, Lin GN, Li M, Xia ZJ. The effect of prophylactic lamivudine on hepatitis B virus reactivation in HBsAg-positive patients with diffuse large B-cell lymphoma undergoing prolonged rituximab therapy. Med Oncol. 2012;29:1237-41.[PubMed Abstract] -
- Esteve M, Saro C, González-Huix F, Suarez F, Forné M, Viver JM. Chronic hepatitis B reactivation following infliximab therapy in Crohn's disease patients: need for primary prophylaxis. Gut. 2004;53:1363-5.[PubMed Abstract] -
- Fung J, Lai CL, Seto WK, Yuen MF. Nucleoside/nucleotide analogues in the treatment of chronic hepatitis B. J Antimicrob Chemother. 2011;66:2715-25.[PubMed Abstract] -
- Hsiao LT, Chiou TJ, Liu JH, et al. Extended lamivudine therapy against hepatitis B virus infection in hematopoietic stem cell transplant recipients. Biol Blood Marrow Transplant. 2006;12:84-94.[PubMed Abstract] -
- Katz LH, Fraser A, Gafter-Gvili A, Leibovici L, Tur-Kaspa R. Lamivudine prevents reactivation of hepatitis B and reduces mortality in immunosuppressed patients: systematic review and meta-analysis. J Viral Hepat. 2008;15:89-102.[PubMed Abstract] -
- Kim PS, Ho GY, Prete PE, Furst DE. Safety and efficacy of abatacept in eight rheumatoid arthritis patients with chronic hepatitis B. Arthritis Care Res (Hoboken). 2012;64:1265-8.[PubMed Abstract] -
- Koukoulioti E, Brodzinski A, Mihm U, et al. Risk factors for resistance development against lamivudine during long-term treatment of chronic hepatitis B virus infections. Eur J Gastroenterol Hepatol. 2019;31:845-52.[PubMed Abstract] -
- Lok AS, Lai CL, Leung N, et al. Long-term safety of lamivudine treatment in patients with chronic hepatitis B. Gastroenterology. 2003;125:1714-22.[PubMed Abstract] -
- Loomba R, Rowley A, Wesley R, et al. Systematic review: the effect of preventive lamivudine on hepatitis B reactivation during chemotherapy. Ann Intern Med. 2008;148:519-28.[PubMed Abstract] -
- Perrillo RP, Gish R, Falck-Ytter YT. American Gastroenterological Association Institute technical review on prevention and treatment of hepatitis B virus reactivation during immunosuppressive drug therapy. Gastroenterology. 2015;148:221-244.e3.[PubMed Abstract] -
- Perrillo RP, Martin P, Lok AS. Preventing hepatitis B reactivation due to immunosuppressive drug treatments. JAMA. 2015;313:1617-8.[PubMed Abstract] -
- Tang W, Chen L, Zheng R, et al. Prophylactic Effect of Lamivudine for Chemotherapy-Induced Hepatitis B Reactivation in Breast Cancer: A Meta-Analysis. PLoS One. 2015;10:e0128673.[PubMed Abstract] -
- Wang ST, Tseng CW, Hsu CW, et al. Reactivation of hepatitis B virus infection in patients with rheumatoid arthritis receiving tofacitinib. Int J Rheum Dis. 2021;24:1362-9.[PubMed Abstract] -
- Weinbaum CM, Williams I, Mast EE, et al. Recommendations for identification and public health management of persons with chronic hepatitis B virus infection. MMWR Recomm Rep. 2008;57:1-20.[PubMed Abstract] -
- Xu Z, Dai W, Wu YT, et al. Prophylactic effect of lamivudine on chemotherapy-induced hepatitis B virus reactivation in patients with solid tumour: A meta-analysis. Eur J Cancer Care (Engl). 2018;27:e12799.[PubMed Abstract] -
Figures

Tables
| Reaction Risk | Recommendation | Implementation Considerations |
| High Risk | Antiviral prophylaxis over monitoring alone. (Strong recommendation, moderate certainty evidence) |
|
| Moderate Risk | Antiviral prophylaxis over monitoring alone. (Conditional recommendation, moderate certainty evidence) |
|
| Low Risk | Monitoring alone over antiviral prophylaxis. (Conditional recommendation, moderate certainty evidence) |
|
| Abbreviations: HBV = hepatitis B virus; ALT = alanine aminotransferase This table is based on recommendations from the AGA Clinical Practice Guideline on the Prevention and Treatment of Hepatitis B Virus Reactivation in At-Risk Individuals. |
||
- Ali FS, Nguyen MH, Hernaez R, et al. AGA Clinical Practice Guideline on the Prevention and Treatment of Hepatitis B Virus Reactivation in At-Risk Individuals. Gastroenterology. 2025;168:267-84. [PubMed Abstract]







